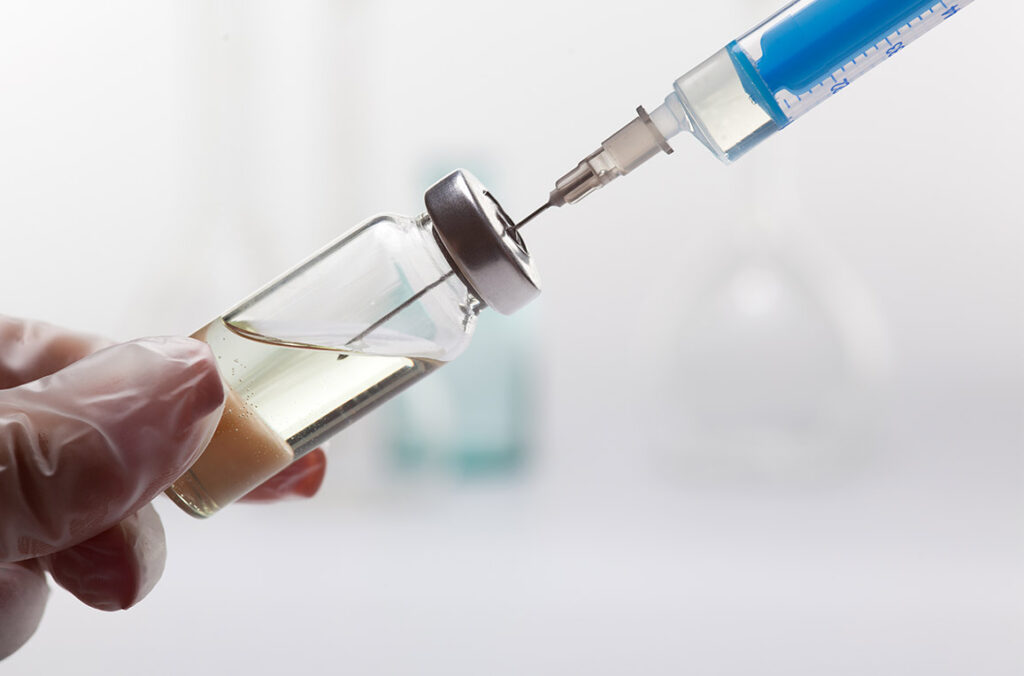
Doctors Center Pharmacy specializes in sterile compounding and is equipped with a state-of-the art ISO Class 7 sterile room that is required for the production of sterile preparations. We have an ISO-Tech ISO Class 5 laminar flow hood which is located in the ISO Class 7 sterile room in which all sterile preparations are made. This set up exceeds the requirements set by the USP 795, 797 guidelines and ensures the highest standards required for sterile production. This level of commitment to our patients is our number one priority resulting in confidence from our customers that our sterile compounds are indeed sterile and safe to inject.
Doctors Center Pharmacy’s laboratory is equipped with a dry heat sterilization oven to sterilize and dehydrogenate all glassware and utensils involved in the compounding process to prevent contamination of bacteria or pyrogens. We also use commercially available sterile and pyrogen free water and saline for injections, vials and filters in all our sterile products to minimize any potential source of contamination.
We have a variety of methods for sterilization available including a dry heat oven and various sterile 0.2-micron filters that enables us to sterilize a variety of products, which all require different methods of sterilization. Our sterile room undergoes daily bactericidal cleaning to maintain a sterile environment. We also monitor the environment by taking air samples to ensure no bacteria are present. We also perform glove fingertip tests to ensure that our compounders have the proper aseptic technique. We also use surface contact plates to sample surfaces weekly that are incubated at the required conditions to ensure the absence of any microbes.
We routinely run our own in-house sterility testing as part of our quality control to ensure our products are sterile. This involves incubating samples in various growth media in an incubator for 14 days to ensure the absence of any microbial growth. We also perform bubble point testing on our sterile filters to validate their integrity which ensures their ability to sterilize heat sensitive mixtures.
LEARN MORE ABOUT OUR AUTOLOGOUS SERUM EYE DROPS
READ MORE ABOUT OUR STERILE PREPARATIONS
Contact our compounding pharmacist at 334-793-1316 or by email for more information.